The practice of storing drinking water in copper vessels has a long history in several cultures. In recent years, it has attracted renewed attention.
This article presents verified, research-based information on copper-stored water, separating scientifically established facts from unsupported claims.

1. Antimicrobial Properties of Copper
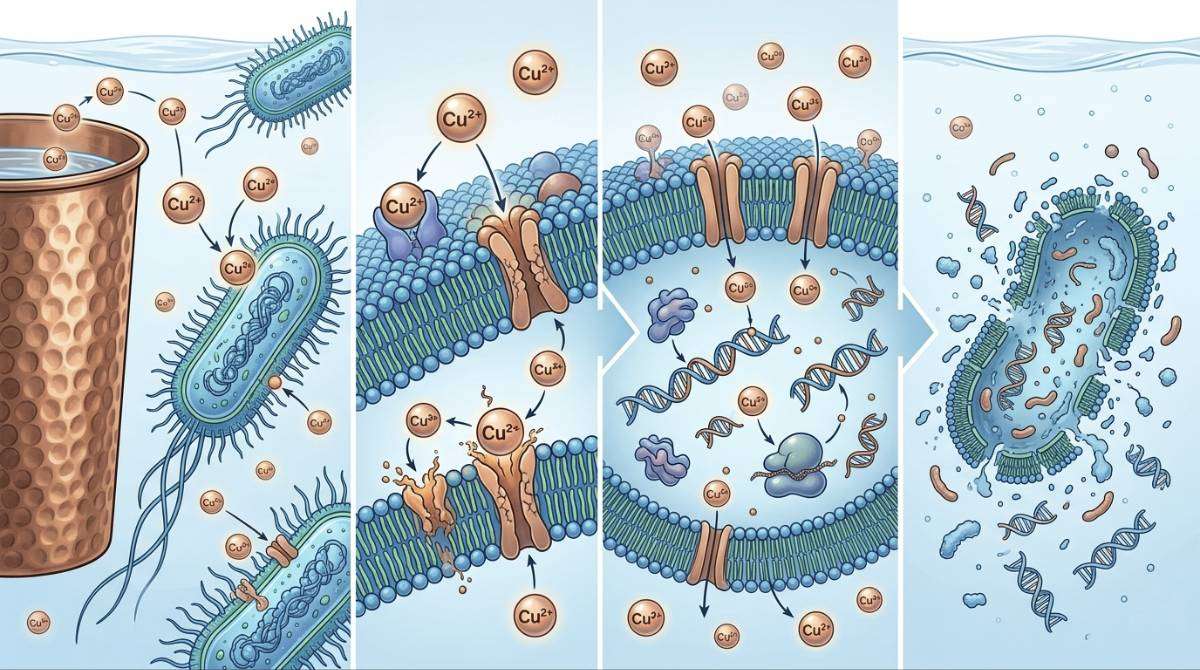
Copper is recognized for its antimicrobial activity. Scientific studies have shown that copper surfaces can inactivate a range of microorganisms. The World Health Organization acknowledges that safe water storage is important for preventing microbial contamination.
Research published in the National Institutes of Health indicates that storing water in copper vessels for several hours can significantly reduce harmful bacteria such as Escherichia coli and Salmonella.
This antimicrobial effect is due to the oligodynamic action, where copper ions disrupt microbial cell membranes and metabolic processes.
2. Release of Copper Ions into Water
When water is stored in copper containers, trace amounts of copper ions dissolve into the water. Copper is classified as an essential trace element required for physiological functions such as enzyme activity and iron metabolism.
According to the World Health Organization, copper intake must remain within safe limits. The concentration of copper in stored water depends on factors such as storage time, temperature, and water chemistry.
3. Safety Standards and Limits
International guidelines define safe limits for copper in drinking water The World Health Organization guideline value for copper in drinking water is approximately 2 mg/L.
The United States Environmental Protection Agency (USEPA) sets a similar action level of 1.3 mg/L for copper in drinking water systems. Excess copper intake may lead to adverse effects, including gastrointestinal discomfort and, in extreme cases, liver or kidney issues.
4. Limitations of Copper Storage
While copper exhibits antimicrobial properties, it is important to understand its limitations Copper does not remove chemical contaminants, including pesticides, heavy metals, or dissolved salts.
It is not a substitute for modern water purification methods such as filtration, reverse osmosis, or ultraviolet treatment. The antimicrobial effect requires sufficient contact time (typically several hours) and is not instantaneous.
5. Scientific Position on Common Claims
Claim: Copper water is the “purest water in the world.”
Claim: Copper water contains a special “electric charge.”
There is no scientific or regulatory evidence supporting this statement. Water purity is determined by comprehensive removal of physical, chemical, and biological contaminants, which copper alone cannot achieve. There is no validated scientific evidence that water stored in copper develops any unique electrical property beneficial to human health.
6. Practical Usage Guidelines
Based on available evidence and safety recommendations Store potable water in a clean copper vessel for 6–12 hours before consumption. Avoid storing acidic liquids (such as citrus juices) in copper containers, as this can increase copper leaching.
Ensure vessels are regularly cleaned to prevent surface contamination. Use copper storage as a supplementary practice, not a replacement for safe water treatment.

READ THIS: Microplastics in Bottled Water: Shocking Facts You Must Know in 2026
Conclusion
Scientific evidence supports that copper vessels can reduce microbial contamination and introduce trace amounts of an essential mineral into drinking water.
However, copper storage does not purify water completely and does not provide any special electrical properties. For safe consumption, it should be used alongside established water treatment methods and with in recommended exposure limits.